Tasting Notes – Acid’s Role in Wine
Acid is the backbone in wine and adds freshness, balance, and structure to wine. It has an impact on everything from taste to aging potential, and food pairings.
A wine’s acidity starts in the vineyard. Unripe grapes have high acid levels, but that drops as they ripen. Grapes grown in cooler climates usually contain higher acidity because there’s less warmth and sunshine available to increase grapes’ sugar and pH levels.
What is pH?
The pH scale measures how acidic or alkaline a substance is. The scale ranges from 0 to 14. A pH of 7 is neutral. A pH less than 7 is acidic, and a pH greater than 7 is basic. Pure water is neutral, with a pH of 7.0. For comparison, acidic white vinegar tends to have a pH level of 2.5, while milk registers at around 6.5, and water clocks in at a neutral 7. Typically, the pH level of a wine ranges from 3 to 4.
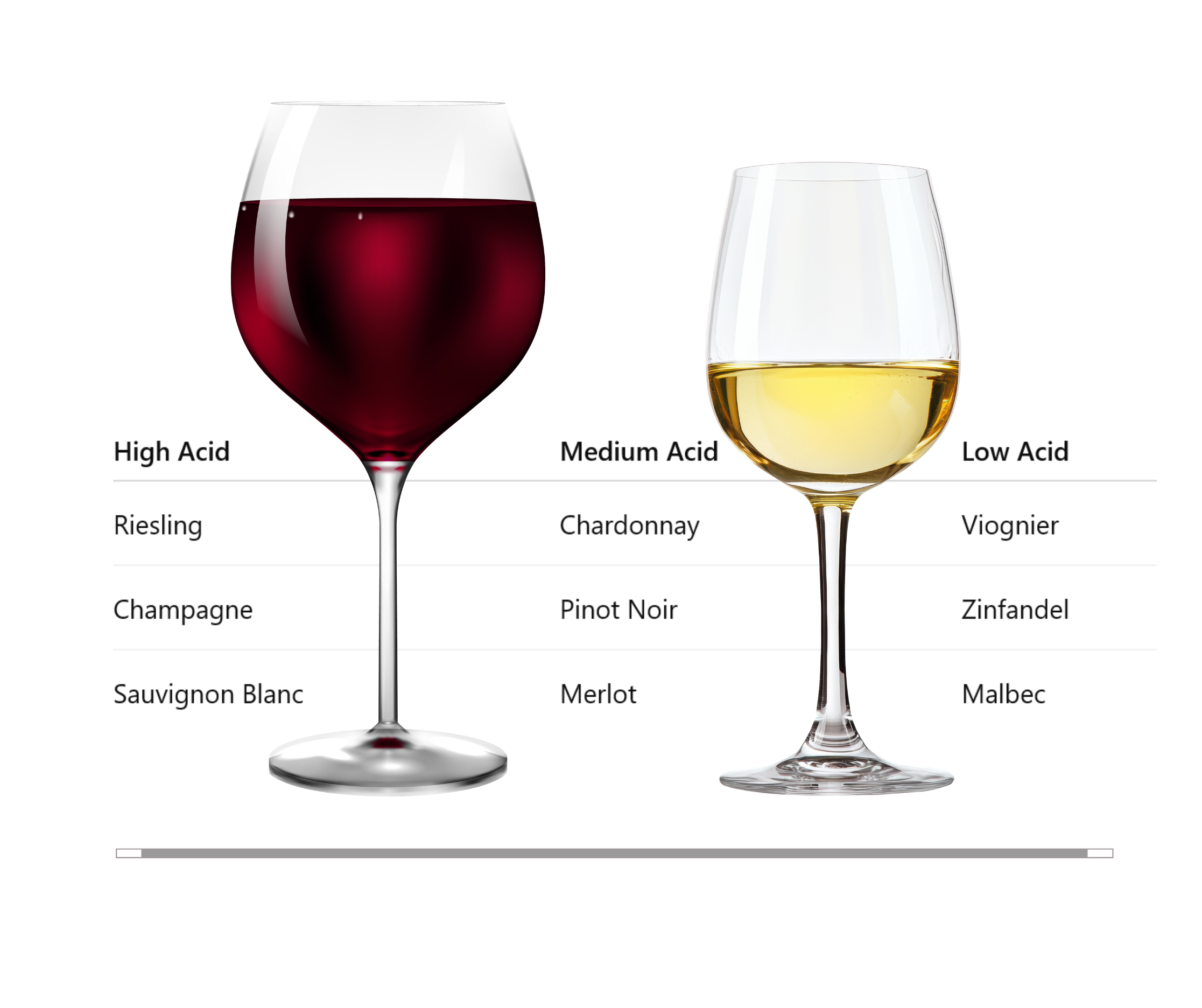
How do I know how much acid is in a specific wine?
Hold your mouth open after you sip. If you begin salivating, your mouth is reacting to the acid. The more saliva, the more acid.
Three Main Acids in Wine
The three main acids in wine are Tartaric Acid, Malic Acid, and Citirc acid. Tartaric Acid is the most prevalent acid in wine and is the key player in maintaining the wine’s chemical stability and color. Malic Acid adds an apple-like freshness to the wine, which can be softened through malolactic fermentation. Citric Acid, the least prevalent acid, adds a touch of freshness and zest.
What Acid Does for Wine
Acid helps balance the other key characteristics of wine – sweetness, alcohol, and tannins. Acid prevents wine from tasting too syrupy. It keeps a wine from seeming too heavy and flat. Along with tannins, acidity adds structure and complexity. Acid helps wine be an incredible complement to food. It’s what makes your mouth water and prepares it for the next bite of food. It also ensures wine’s ability to age and stay fresh in the glass.